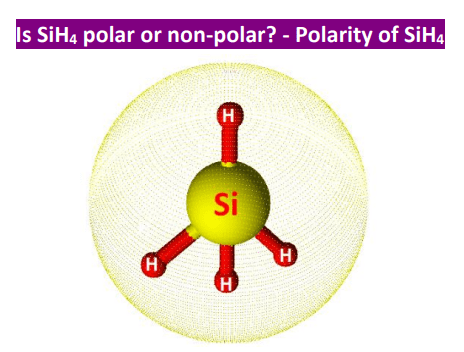
SOLVED: Draw the Lewis Structure for SiH4 to decide whether it's polar or nonpolar. A nonpolar B polar

SOLVED: Choose the selection which correctly characterizes all three of the following substances in terms of whether they are polar or nonpolar: SiH4 and BBr3 and SiF4 a) SiH4 is nonpolar and